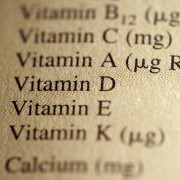 Photo: Getty Images
Photo: Getty Images
Some people choose to use vitamin pills and other supplements to help fill in the gaps and give their bodies the vitamins that are not provided by the foods they eat. Because vitamins are sold over-the-counter at grocery stores, pharmacies, and health food stores, it may be natural to assume that all these products have been tested and are safe for anyone who wants to take them. This is not the case. Supplements do not have the same degree of regulation in the United States as prescription drugs or foods. Some supplements are of better quality than others and provide more valuable nutrients.
Before you chose a supplement, compare the labels to make sure you are really getting what you want. The U.S. Food and Drug Administration regulates three different kinds of claims made on supplement labels to ensure they are accurate:
• Health claims – Supplements may claim to reduce the risk of a disease or other health-related condition based on the ingredients in the supplement.
• Nutrient claims – Supplement labels often claim to have a certain amount of a nutrient or other substance.
• Function/structure claims – Supplements may claim to affect certain systems of the body including certain organs. Function/structure claims cannot mention preventing or curing a specific disease.
It is important to note that the FDA assumes supplement ingredients that have been on the market prior to October 15, 1994 are safe based on their history of use by humans. These ingredients can be marketed without further review by the FDA. New ingredients (those not in use prior to October 15, 1994) must be approved by the FDA before they can be marketed as supplements.
The FDA requires supplements to have accurate labels in order to be sold for human consumption. This information must be included on the label:
• Name of product (must also indicate that the product is a supplement)
• Net quantity of contents
• Name and location of the business that is the manufacturer, packer, or distributor
• Instructions for taking the supplement
Before you start taking supplements including vitamins, it is important to know how much of that supplement your body needs each day. Reading the labels on vitamin bottles can be confusing unless you understand some of the abbreviations used.
• % Daily Value or %DV - In the United States the Institute of Medicine, which is a private organization located in Washington, D.C., sets the daily Recommended Dietary Allowance (RDA) for many vitamins and minerals. The percent listed is the percent of that daily value contained in one serving of the supplement. For example, if the line for vitamin A shows a %DV as 45%, you will receive 45 percent of the daily recommended dose of Vitamin A when you take one serving of the supplement.
For some substances, especially minerals such as nickel or tin, you may see an asterisk (*) in place of a percent value. This is to show that the Institute of Medicine has not established guidelines for the daily recommended dose, but the minerals are included in the supplement because they are known to be necessary for healthy body function.
• Serving size - This shows how many pills, capsules, or tablets equals one serving. You need to take this much to get the %DV listed for each nutrient on the label. Don’t assume that one tablet is the serving, or that all supplements are intended to be taken once a day. Read the label to make sure you are getting the full benefit from the supplement and that you are not taking too much.
• Scientific units – Some vitamins are shown as a number of milligrams (mg) or micrograms (mcg). Others may say some number of International Units (IU). These are all ways supplements can be measured. In general, fat –soluble vitamins and minerals such as vitamins A, D, and E are measures in International Units. Water-soluble vitamins such as the B vitamins and vitamin C are measure in fractions of a gram (milligrams or micrograms).
Other important information you will find on supplement labels include:
• Expiration date – supplements that are older than the listed date will not provide the full daily dose listed on the label.
• Lot number – This acts as a tracking number in case there are any concerns about the product batch such as contamination with other substances or any other health concerns.
• Dosing instructions – Manufacturers may provide suggestions for when to take the supplement to get the most benefit, such as taking on an empty stomach or at bedtime.
• Quality statements – reputable manufacturers often include a statement of quality or a seal from a health group that provides an endorsement for the product. Examples include a statement from the United States Pharmacopoeia (USP) or a medical association seal.
Before adding any new supplements to your diet, talk to you doctor to make sure the supplement will benefit your health and will not conflict with any other medications you are taking for any health concerns.
Sources:
National Institutes of Health Office of Dietary Supplements
About.com: Understanding Vitamin and Herb Labels






Add a CommentComments
There are no comments yet. Be the first one and get the conversation started!