 iStockphoto/Thinkstock
iStockphoto/Thinkstock
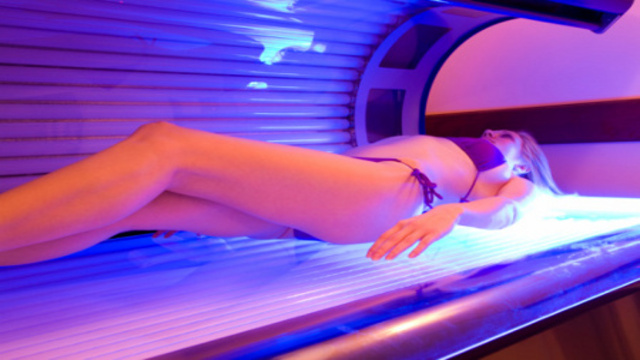
Tanning beds and sun lamps are targeted by the Food and Drug Administration (FDA) in its new proposed legislation that requires those devices to be reclassified as “moderate risk” instead of “low risk”. In addition, they both will be required to have stronger warning labels that people under the age of 18 should not use them.
The FDA wants to change the current classification of tanning beds in order to have stricter standards to regulate them. They claim that faulty equipment has led to patients being burned or exposed to too much radiation, according to Medscape.com.
The FDA states that the proposed guidelines will require premarket approval of tanning beds and sun lamps before they can be sold to the public. Current guidelines do not require this.
However, the suggested revisions are not as strict as the laws in many states. The Environmental Work Group (EWG) states on their website that “the FDA should take a stronger stand against indoor tanning beds, particularly for teens.”
According to EWG, 33 states restrict teen access to tanning beds. Medscape reported that Delaware, New Hampshire and North Dakota ban the use of indoor tanning by anyone under the age of 14 unless needed for medical reasons. Parental consent is needed for those aged 14-18.
The New York Times reported that as of January 2012, California became the first state to ban the use of tanning beds for all people under the age of 18.
The FDA panel did not approve a move to ban tanning beds. The panel was split on the level of strictness the level of classification change, but all agreed a change was needed. Only a few members of the panel suggested an outright ban of tanning beds.
It is curious that the panel did not have more members who wanted stronger laws.
The FDA press release stated that, “according to the American Academy of Dermatology, there is a 75 percent increase in the risk of melanoma, the deadliest type of skin cancer, in those who have been exposed to ultraviolet radiation from indoor tanning, and the risk increases with each use.”
They went on to say, “The proposed order does not prohibit the use of sunlamp products by those under the age of 18, but it provides a warning on the consequences.”
EWG thinks that the FDA should state outright that use of “tanning beds causes skin cancer” not just make a mild statement that those under 18 should not use the devices.
According to EWG, even the FDA’s own regulations note that melanoma is the second most common type of cancer in young adults and many experts believe that use of sun lamps has contributed to this statistic.
EWG recommends that overall, people should not use tanning beds or sunbathe outdoors. People should make every attempt to avoid getting sunburned.
Wearing sunscreen is not enough protection because it allows people to stay out longer in the sun and be exposed to more UV rays.
EWG goes on to warn consumers, if you have used tanning beds in the past or have a history of sunburns, be especially diligent in checking your skin for new eruptions or changes in any skin lesion.
The FDA has encouraged consumers to comment on the proposed regulations until August 7, 2013. You can go to this link and type in “tanning beds” in the search line to pull up a page with the proposed regulation. Click on the “comment now” button on the right.
Sources:
Reuters Health Information: FDA Strengthens Warnings on Tanning Beds. Medscape Nurses News. Retrieved June 3, 2013.
http://www.medscape.com/viewarticle/803691?nlid=31248_1587
F.D.A. Proposes New Tanning Bed Warnings. New York Times. Retrieved June 3, 2013.
http://well.blogs.nytimes.com/2013/05/06/f-d-a-proposes-new-tanning-bed-...
FDA Proposes Warning Labels On Tanning Beds. Environmental Working Group. Enviroblog. Retrieved June 3, 2013.
http://www.ewg.org/enviroblog/2013/05/fda-action-tanning-beds
FDA NEWS RELEASE: FDA issues proposal to increase consumer awareness of tanning bed risks. U.S. Food and Drug Administration. Retrieved June 3, 2013.
http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm350864.htm
Sunbeds, tanning and UV exposure. World Health Organization. Retrieved June 3, 2013.
http://www.who.int/mediacentre/factsheets/fs287/en/
Michele is an R.N. freelance writer with a special interest in woman’s healthcare and quality of care issues. Other articles by Michele are at www.helium.com/users/487540/show_articles
Edited by Jody Smith



Add a CommentComments
There are no comments yet. Be the first one and get the conversation started!