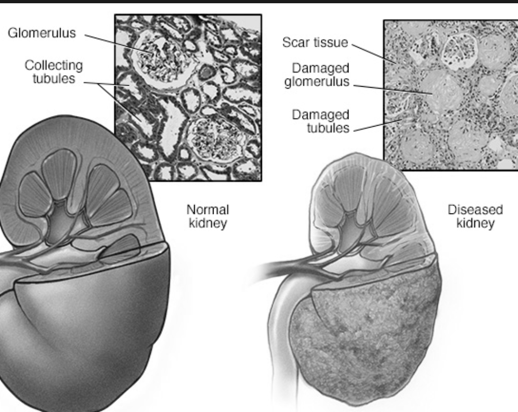
The list of individual drugs that cause kidney damage is so long that it's hard to decide on the top ten. So I've chosen to list them by type of drug: antibiotic, analgesic, etc.
Top ten drugs that cause kidney damage:
1. Antibiotics, including ciprofloxacin, methicillin, vancomycin, sulfonamides.
2. Analgesics, including acetominophen and non-steroidal anti-inflammatory drugs (NSAID): aspirin, ibuprofen, naproxen, and others available only by prescription.
4. Heartburn drugs of the proton pump inhibitor class, including omeprazole (brand name Prilosec), lansoprazole (brand name Prevacid), pantoprazole (brand name Protonix), rabeprazol (brand names Rabecid, Aciphex), esomeprazole (brand names Nexium, Esotrex). See Ref. 4.
5. Antiviral drugs, including acyclovir (brand name Zovirax) used to treat herpes infection, and indinavir and tenofovir, both used to treat HIV.
6. High blood pressure drugs, including captopril (brand name Capoten).
7. Rheumatoid arthritis drugs, including infliximab (brand name Remicade); chloroquine and hydroxychloroquine, which are used to treat malaria and systemic lupus erythematosus as well as rheumatoid arthritis.
8. Lithium, used to treat bipolar disorder.
9. Anticonvulsants, including phenytoin (brand name Dilantin) and trimethadione (brand name Tridione), used to treat seizures and other conditions.
10. Chemotherapy drugs, including interferons, pamidronate, cisplatin, carboplatin, cyclosporine, tacrolimus, quinine, mitomycin C, bevacizumab; and anti-thyroid drugs, including propylthiouracil, used to treat overactive thyroid.
For many prescription drugs, you can find the full prescribing information online. This information is typically 10 – 30 pages long, and includes detailed precautions, warnings, and adverse effects. To find it, do a search for the name of your drug. Find the web site provided by the manufacturer. In most cases, it's just the brand name of the drug with a “.com” added. Then find the link to “prescribing information” and click on that.
For over-the-counter drugs, be sure to observe the precautions on the label. Keep your doctor informed of what you're taking, especially if you have risk factors for kidney disease (see Ref. 2) or other health conditions.
by Linda Fugate, Ph.D.
References:
1. Loh AHL, Cohen AH, “Drug-induced Kidney Disease – Pathology and Current Concepts”, Ann Acad Med Singapore 2009;38:240-50.
2. https://www.empowher.com/news/herarticle/2009/09/02/top-ten-risk-factors-chronic-kidney-disease
3. https://www.empowher.com/news/herarticle/2009/10/26/what-nsaids-do-your-kidneys
4. https://www.empowher.com/news/herarticle/2009/09/09/rare-serious-side-effect-heartburn-drugs
Linda Fugate is a scientist and writer in Austin, Texas. She has a Ph.D. in Physics and an M.S. in Macromolecular Science and Engineering. Her background includes academic and industrial research in materials science. She currently writes song lyrics and health articles.



Add a Comment15 Comments
I take omeprazole for reflux, went to one kidney dr. said that was causing kidney problems.
June 20, 2018 - 5:53amThis Comment
Laprazol fast tabs caused me severe pain in my kidneys, back pain and spinal pain. It is so painful that I cannot even walk or sleep at night. The only way to overcome the pain is by taking depon pills which alleviates the pain for only 4 hours
August 28, 2015 - 6:07pmThis Comment
I would like to know where the good doctor got her information. Some of her info I would have to question. I have systemic lupus and I see the top rheumatologist in the country and he prescribes hydroxychloroquine (Plaquenil) for his lupus patients. This is pretty much a miracle drug for lupus patients. It helps with all the joint pains, fevers, keeps cholesterol down, and prevents kidney damage. The only rare problem involves some loss of vision, but again, very rare and not an issue if you see an eye doc every six months to catch problem early. I really would like to know why she included this drug. She doesn't list her sources, so we can't really gage how accurate this info is.
April 5, 2015 - 2:32amThis Comment
Have you even read the side effects to that drug, or are you only listening to what your doctor has to say about it? Here are the full list of side effects for hydroxychloroquine - oral, Plaquenil... and it surely isn't just the vision that could be affected by taking this drug: SIDE EFFECTS: Nausea, stomach cramps, loss of appetite, diarrhea, dizziness, or headache may occur. If any of these effects persist or worsen, notify your doctor or pharmacist promptly.Remember that your doctor has prescribed this medication because he or she has judged that the benefit to you is greater than the risk of side effects. Many people using this medication do not have serious side effects.Tell your doctor immediately if any of these unlikely but serious side effects occur: arm/leg/back pain, fast heartbeat, hair loss/color change, mental/mood changes (e.g., anxiety, depression, hallucinations), ringing in the ears/hearing loss, worsening of skin conditions (e.g., psoriasis).This medication may infrequently cause serious (sometimes permanent) eye problems or muscle damage, especially if you take it for a long time. Seek immediate medical attention if any of these unlikely but very serious side effects occur: sensitivity to light, vision changes (e.g., blurred vision, seeing light flashes/streaks/halos, missing/blacked-out areas of vision), muscle weakness.Tell your doctor immediately if any of these rare but very serious side effects occur: severe stomach/abdominal pain, severe nausea/vomiting, easy bleeding/bruising, signs of infection (e.g., fever, persistent sore throat), seizures, shortness of breath, swelling ankles/feet, extreme tiredness, dark urine, yellowing eyes/skin.A very serious allergic reaction is unlikely, but seek immediate medical attention if it occurs. Symptoms of a serious allergic reaction may include: rash, itching/swelling (especially of the face/tongue/throat), dizziness, trouble breathing.This is not a complete list of possible side effects. If you notice other effects not listed above, contact your doctor or pharmacist. https://www.medicinenet.com/hydroxychloroquine-oral/article.htm
July 21, 2019 - 12:00pmThis Comment
I have kidney disease and RA and melanoma I was recently put on plaque nil 200mgs and have had nausea diarrea skin rash dizziness headaches and now severe pain in my right side and kidney. My pharmacist warned me to be cautious about taking this drug she was right now I'm having to take more antibiotics for the infection in my kidney. Bad drug not enough info regarding kidney disease and taking this drug
June 16, 2016 - 8:36amThis Comment
I noticed Tylenol is on the list and it is actually recommended by the national kidney foundation.
June 5, 2015 - 5:17pmThis Comment
Yes... I agree! Tylenol is metabolized by the liver, so how is that dangerous for the kidneys??? Now these NSAIDS on the other hand are metabolized by the kidneys, so that one small thing you would be correct on, but I can give you a 99.9% guarantee that taking Tylenol will not damage your kidneys and is the preferred choice actually!!!
April 28, 2017 - 7:21pmThis Comment
I took 40 mg of Prilosec every day for a year - stomach ulcers. Now my urine is foamy and I have proteinuria. 36 white male, used to be healthy. Oh well. I guess I threw my life away. Thanks, Prilosec.
February 16, 2015 - 2:12pmThis Comment
Hi,
Thanks for this, understanding and looking after your Kidneys, as a key organ, is very important. If you're concerned check out the symptoms here:
http://www.patient.co.uk/health/mild-to-moderate-chronic-kidney-disease
Hope this helps.
February 12, 2013 - 4:11amThis Comment
I have recently been diagnosed with stage 2 ckd. I have been on Nexium (mostly 2 times a day) since 1999. Could the Nexium have attributed to my ckd? I do have high blood pressure and was told this is the cause of my ckd.
March 30, 2010 - 12:48pmThis Comment