Certain drugs used to prevent rejection of transplanted kidneys must carry a warning about the risk of serious infections, the U.S. Food and Drug Administration announced Tuesday.
The drugs include CellCept (mycophenolate mofetil) and generics, Myfortic (mycophenolic acid), Rapamune (sirolimus), Sandimmune (cyclosporine) and generics, and Neoral (cyclosporine modified) and generics.
The drugs, which already carry the FDA's most serious boxed warning outlining their various risks, must now also mention the increased risk of "opportunistic infections," including activation of dormant viral infections such as one caused by the BK virus, the Dow Jones news service reported.
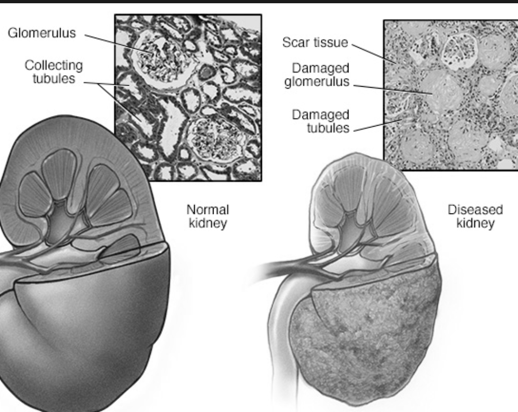
Infections associated with the immunosuppressive drugs may lead to serious problems, including kidney graft loss, said the FDA. The agency noted that a warning about the increased risk for opportunistic infections already is included in the labeling of the immunosuppressive drug Prograf (tacrolimus).